Research from the Schnöbrunn laboratory focuses on the elucidation of the structure-activity relationship of medicinally important proteins. We use protein crystallography combined with methods in medicinal chemistry, biochemistry and molecular biology to explore proteins at the atomic level. Our aim is to identify "weak sites" in those proteins that can be targeted by new inhibitors with potential as future drugs.
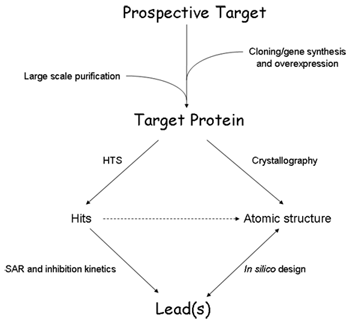
The flowchart details the diversity of methods and techniques being applied in our laboratory for the discovery of novel inhibitors.
Once we have identified a prospective drug target, the first step is the cloning or chemical synthesis of the gene, followed by overexpression of the gene product in suitable host cells and purification of the target protein in the quantities required for the drug discovery enterprise. We generally follow two routes towards the discovery of novel inhibitors. Both these approaches become interconnected if the target protein can be crystallized.
The empirical approach involves the development of an assay suitable for high-throughput screening (HTS) of hundreds of thousands of small organic compounds for inhibitory activity. Thus discovered inhibitors (hits) will be scrutinized by structure-activity relationship (SAR) and kinetic studies until the most potent inhibitors with drug-like properties (leads) have been identified.
The second route is the rational design of inhibitors based on the 3D atomic structure of the target protein. First, crystallization conditions suitable for reproducible growth of X-ray quality crystals need to be established and the atomic structure of the target protein will be solved by crystallographic methods, bound with ligands such as substrates, known inhibitors or newly discovered HTS hits and leads. With this information in hand, we perform several computational studies (in silico design), such as molecular docking, to identify chemical scaffolds that satisfy the criteria for high inhibitory potential.
During the entire inhibitor discovery process we closely collaborate with researchers of various disciplines, from synthetic organic chemistry to cell biology, to devise strategies for the optimization of the best inhibitors with respect to drug-like properties. Our expertise enables us to not only thoroughly characterize the molecular mode of action of inhibitors on proteins; we also perform mechanistic studies, for example to trap reaction intermediate states of enzyme-catalyzed reactions. Furthermore, we investigate the resistance of target proteins to known inhibitors. These studies complement the rational design approach, at the same time providing valuable information about the relationship of the proteins structure and function.