(Presented as a poster at the 2023 American Association for Cancer Research (AACR) Annual Meeting)
Authors:
John Charles A. Lacson, MS, PhD1*, Youngchul Kim, PhD2, Richard G. Roetzheim, MD3, Steven K. Sutton, MS, PhD2, Susan T. Vadaparampil, MPH, MS, PhD4, Brenda Soto-Torres, MPHE, PhD5, Peter A. Kanetsky, MPH, PhD1
Affiliations:
1 Department of Cancer Epidemiology, H. Lee Moffitt Cancer Center & Research Institute, Tampa, FL, US
2 Department of Biostatistics and Bioinformatics, H. Lee Moffitt Cancer Center and Research Institute, Tampa, FL, US
3 Department of Family Medicine, Morsani College of Medicine, University of South Florida, Tampa, FL, US
4 Department of Health and Behavioral Outcomes, H. Lee Moffitt Cancer Center and Research Institute, Tampa, FL, US
5 Public Health Program, Ponce Health Sciences University, Ponce, Puerto Rico, USA
* Poster presenter, email: JohnCharles.Lacson@moffitt.org
Introduction
- Hispanics are underserved in precision medicine – they are less likely to get referred to and to undergo genetic testing and are more likely to have variants of unknown significance1-5.
- Although Hispanics generally have low awareness of genetic testing services1, those who are aware of such testing perceive several benefits from and report positive attitudes about testing6-8.
- Modest successes in improving preventive behavior, particularly for skin cancer prevention, have been observed from precision prevention trials among both Hispanic and non-Hispanic participants that directly provided genetic testing results together with genetics-based prevention guidelines to participants9-12.
- Recall is a fundamental psychological mechanism underlying how people receive and act upon health recommendations; better recall is associated with greater adherence to treatment and improved health outcomes13,14.
- There have been no studies that describe recall of genetic testing results among Hispanics.
- Genetic variants in MC1R are markers of susceptibility to skin cancer, including basal cell carcinoma, squamous cell carcinoma, and melanoma, among individuals of European ancestry15,16, including Hispanics and Puerto Ricans specifically17.
- Hypotheses:
- Participants at higher risk are more likely to correctly recall their MC1R risk compared to participants at average risk.
- Better recall at the 3-month follow-up than the 9-month follow-up because of time-related memory decay18.
Materials and Methods
This study reports secondary analyses of data from a previously published randomized controlled intervention trial12.
Participants and setting
- Self-identified Hispanic participants were recruited from 8 primary care clinics and community health centers in Tampa, Florida and Ponce, Puerto Rico from September 2018 to January 2020.
- At least 18 years (Tampa) or 21 years (Ponce) of age.
- Exclusion criteria: having a full-body skin examination within the past year, a previous diagnosis of melanoma, or two or more diagnoses of non-melanoma skin cancer.
- Both English- and Spanish-language study materials were available for participants.
- Bilingual and bicultural research staff were available to interact with participants.
- After providing written informed consent.
- Participants provided a saliva sample for DNA isolation and MC1R sequencing, completed a baseline questionnaire, and were randomized in 1:1 ratio within MC1R risk stratum (average or higher) to receive precision prevention or generic prevention materials (standard).
Precision prevention materials
- Intervention arm: precision prevention materials with participant’s MC1R risk category (average or higher risk), genetics-based prevention guidelines, facts and figures about skin cancer.
- Carriage of specific MC1R variant(s) was not provided.
- Standard arm: generic prevention materials (without genetic information) and skin exam guidelines adapted from the American Academy of Dermatology (AAD)19.
Questionnaire assessments
- Baseline questionnaire (completed before intervention materials were provided) collected information on age, sex, marital status, education, family history of basal cell carcinoma and/or squamous cell carcinoma, family history of melanoma, and family history of other cancers.
- The following measures, which have been described in detail previously12,20, were also assessed: health numeracy, health literacy, absolute and comparative chance of getting skin cancer (including melanoma, squamous cell carcinoma, and basal cell carcinoma), recent concern and worry about skin cancer, perceived severity, self-efficacy, response efficacy, skin cancer worry , and recent distress about skin cancer.
- Supplemental questionnaire, answered by 80% of participants, collected information on pigmentation characteristics, cancer fatalism21, and familism22.
- Baseline questionnaire also assessed 11 prevention activities over the past 12 months via a standardized survey of sun exposure and sun protection behaviors23:
- frequency (never, rarely, sometimes, often, always) of each of five sun protection behaviors:
- 1) using sunscreen
- 2) wearing sunglasses
- 3) seeking shade or using an umbrella while outside
- 4) wearing a shirt with sleeves
- 5) wearing a hat
- 6) number of red or painful sunburns
- 7) frequency (never, rarely, sometimes, often, always) of intentional outdoor tanning
- 8) number of intentional indoor tanning occurrences
- 9) number of hours spent outside between 10 a.m. and 4 p.m. separately for weekdays and weekends
- 10) number of skin examinations performed by oneself or partner
- 11) skin examination (yes/no) performed by a health provider.
- At 3- and 9-months post-intervention, these 11 prevention activities were reassessed to be used as outcomes to estimate intervention efficacy.
- At 3- and 9-months post-intervention, participants in the intervention arm were asked to recall their MC1R genetic risk category (“Yes, it was average risk”; “Yes, it was high risk”; or “No, I don’t recall”).
Statistical analyses
Predictors of risk recall
- Baseline measures of pigmentation phenotypes, demographics, family history of skin and cancers, psychosocial constructs, and prevention activities, and the completion of the telephone follow-up were considered as potential predictors of risk recall.
- Risk recall was modeled as a binary variable using logistic regression.
- Due to small numbers, those who misremembered their genetic risk were grouped with those who did not recall.
- The univariate association with each potential predictor was assessed using appropriate statistical tests (chi-square, t-, Wilcoxon rank sum, Kruskal-Wallis, and ANOVA F- tests), separately for recall at 3 and 9 months after baseline.
- Separately for each MC1R risk category, a backwards stepwise regression was conducted on variables with p<0.20 to build a multivariate logistic regression model.
- A final parsimonious model was selected by minimizing the Akaike Information Criterion (AIC), which optimizes model fit rather than relying on a specified statistical threshold to select individual predictors24.
- MASS and radiant packages in R software (ver 4.1.0, R Foundation for Statistical Computing, Vienna, Austria, RRID:SCR_001905) and RStudio (ver 1.4.1717, RStudio Team, Boston, MA, RRID:SCR_000432)
Assessment of intervention effects
- Intervention effects were reassessed for all trial participants (n=920) using previously published methods11, stratified by MC1R risk category and risk recall reported at 3 months.
- Generalized estimating equations (GEE) to determine the intervention effect on each primary prevention activity measured at 3 and 9 months.
- Adjusted for the baseline measure of the outcome, predictors of outcome, imbalanced baseline variables and predictors of missingness for return of the 3- and 9-month assessments.
- We summed the number of sun protection behaviors practiced often or always and treated this as an additional outcome (assumed to have a normal distribution).
- Each of the five sun protection behaviors were examined as binary outcomes (often or always vs. sometimes, rarely, or never) and were modelled using a logit link function.
- All GEEs used an unstructured correlation relationship of repeated measurements.
- Derived binary variables to indicate ever having a skin examination over the study period, separately for those conducted by a health professional or by self/partner.
- Multivariate logistic regression to estimate ORs stratified by recall at 3 months.
- Participants who reported having a skin exam at baseline and those who did not return their 9-month survey were excluded from these analyses, precluding inclusion of missingness predictors.
- SAS ver. 9.4 (SAS Institute, Cary, NC, USA, RRID:SCR_008567)
Results
- At 9 months, participants at higher risk were three times less likely to correctly recall versus misremember (OR=0.31, 95%CI:0.10-0.95).
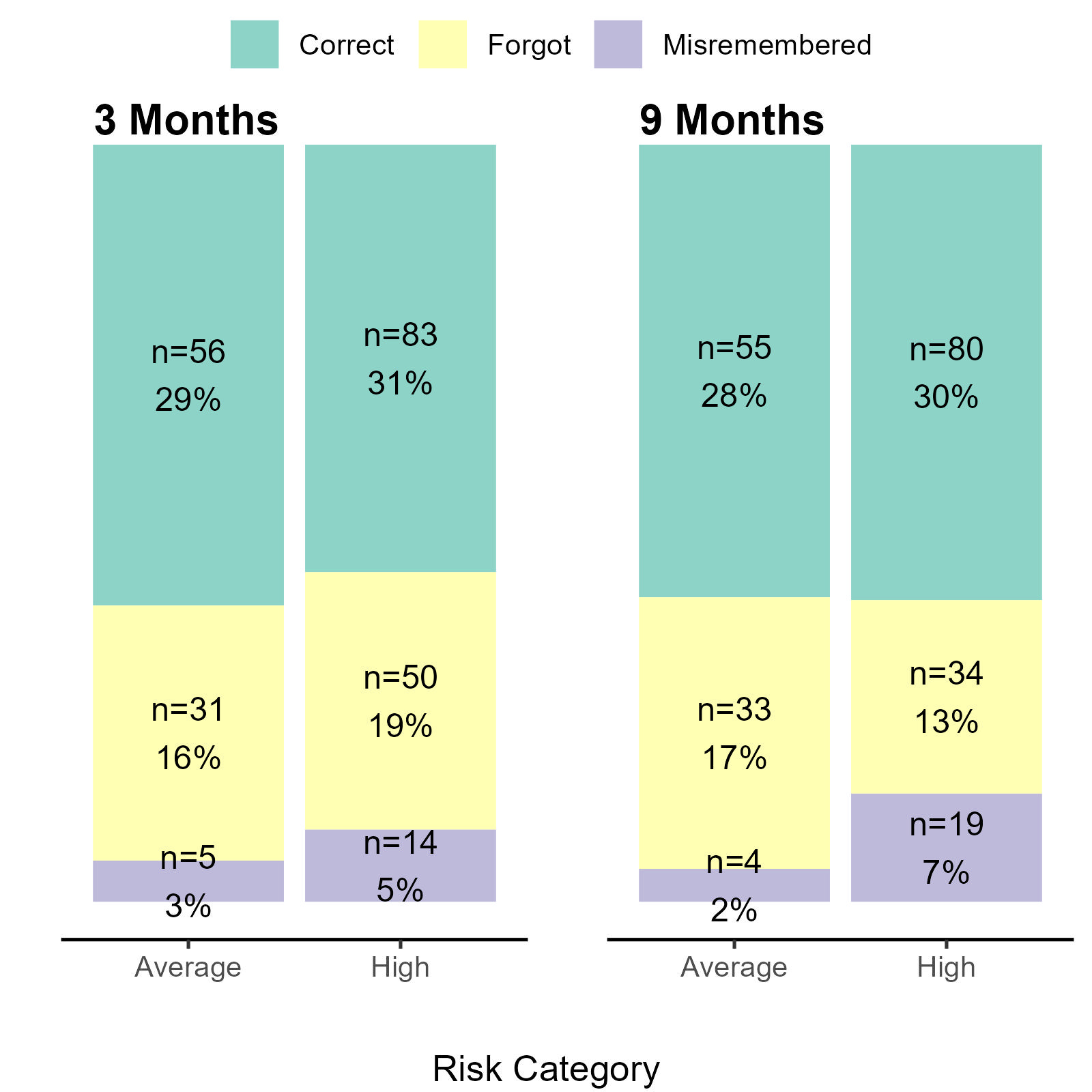
|
Table 1. Baseline predictors (p<0.05) of correct risk recall common across the 3- and 9-month follow-ups.
|
|
Predictor
|
Effect
|
|
MC1R average risk
|
|
Age
|
--
|
|
Education
|
++
|
|
MC1R higher risk
|
|
Education
|
++
|
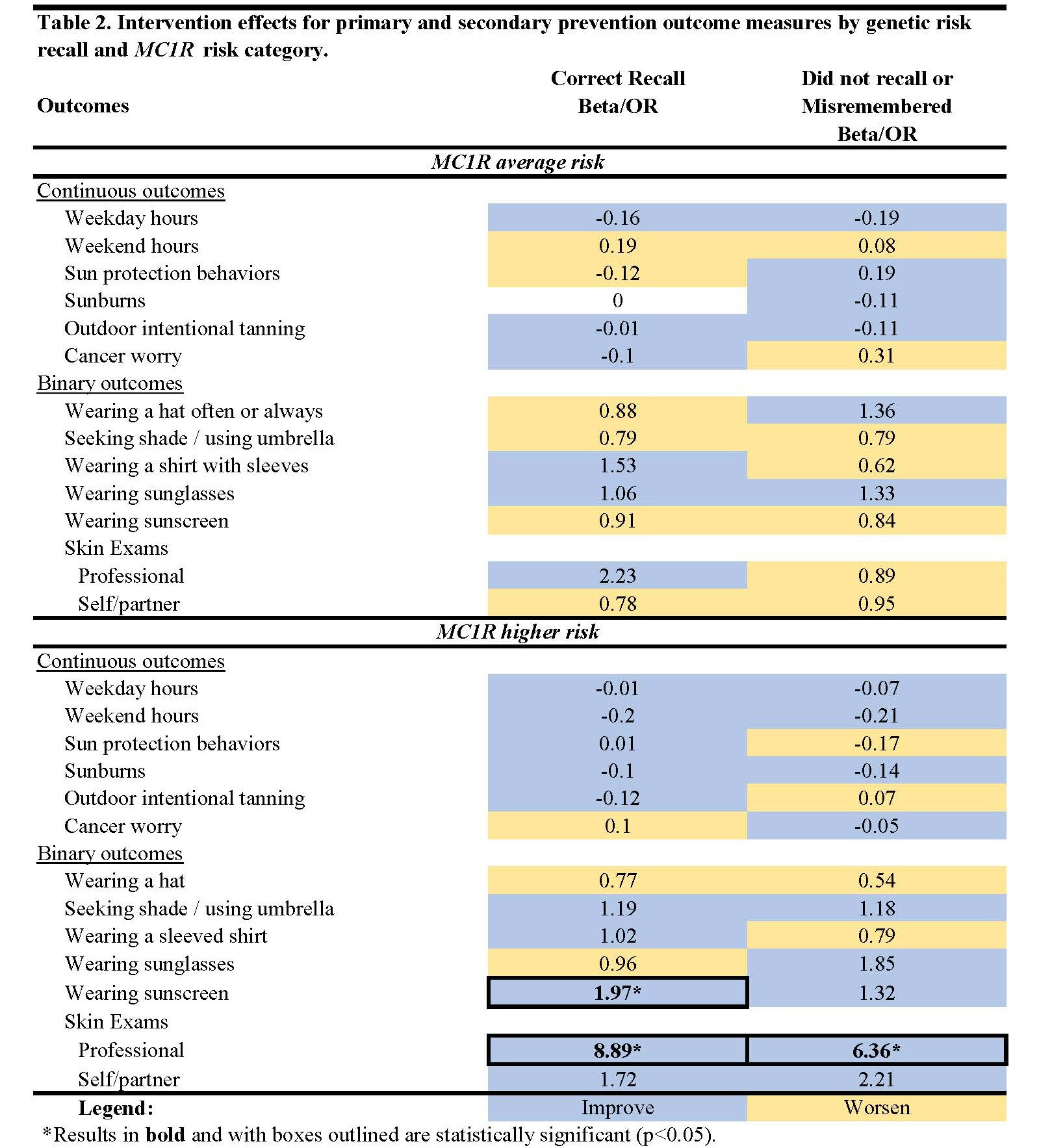
Conclusions
- Consistent with previous studies, participants at higher risk were less likely to correctly recall than participants at average risk.
- Higher educational attainment was a consistent predictor of correct recall among both participants at average and higher risk
- Future studies to further minimize literacy and numeracy demands of their intervention materials and perhaps provide additional support to individuals with lower educational attainment.
- Lastly, findings presented here and in our previous study of recall among non-Hispanic Whites suggest that those who correctly recalled had better intervention outcomes than those who did not recall or misremembered.
References
- Aviles-Santa ML, Heintzman J, Lindberg NM, et al. Personalized medicine and Hispanic health: improving health outcomes and reducing health disparities - a National Heart, Lung, and Blood Institute workshop report. BMC Proc. 2017;11(Suppl 11):11.
- Muller C, Lee SM, Barge W, et al. Low Referral Rate for Genetic Testing in Racially and Ethnically Diverse Patients Despite Universal Colorectal Cancer Screening. Clin Gastroenterol Hepatol. 2018;16(12):1911-1918 e1912.
- Chapman-Davis E, Zhou ZN, Fields JC, et al. Racial and Ethnic Disparities in Genetic Testing at a Hereditary Breast and Ovarian Cancer Center. J Gen Intern Med. 2021;36(1):35-42.
- Martin AR, Kanai M, Kamatani Y, Okada Y, Neale BM, Daly MJ. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat Genet. 2019;51(4):584-591.
- Soewito S, Wyatt R, Berenson E, et al. Disparities in Cancer Genetic Testing and Variants of Uncertain Significance in the Hispanic Population of South Texas. JCO Oncol Pract. 2022;18(5):e805-e813.
- Hann KEJ, Freeman M, Fraser L, et al. Awareness, knowledge, perceptions, and attitudes towards genetic testing for cancer risk among ethnic minority groups: a systematic review. BMC Public Health. 2017;17(1):503.
- Vadaparampil ST, Wideroff L, Breen N, Trapido E. The impact of acculturation on awareness of genetic testing for increased cancer risk among Hispanics in the year 2000 National Health Interview Survey. Cancer Epidemiol Biomarkers Prev. 2006;15(4):618-623.
- Hamilton JG, Shuk E, Arniella G, et al. Genetic Testing Awareness and Attitudes among Latinos: Exploring Shared Perceptions and Gender-Based Differences. Public Health Genomics. 2016;19(1):34-46.
- Frieser MJ, Wilson S, Vrieze S. Behavioral impact of return of genetic test results for complex disease: Systematic review and meta-analysis. Health Psychol. 2018;37(12):1134-1144.
- Smit AK, Allen M, Beswick B, et al. Impact of personal genomic risk information on melanoma prevention behaviors and psychological outcomes: a randomized controlled trial. Genet Med. 2021.
- Lacson JCA, Doyle SH, Qian L, et al. A Randomized Trial of Precision Prevention Materials to Improve Primary and Secondary Melanoma Prevention Activities among Individuals with Limited Melanoma Risk Phenotypes. Cancers (Basel). 2021;13(13):3143.
- Lacson JCA, Doyle SH, Del Rio J, et al. A randomized clinical trial of precision prevention materials incorporating MC1R genetic risk to improve skin cancer prevention activities among Hispanics. Cancer Res Commun. 2022;2(1):28-38.
- Martin LR, Williams SL, Haskard KB, Dimatteo MR. The challenge of patient adherence. Ther Clin Risk Manag. 2005;1(3):189-199.
- McDonald J, McDonald P, Hughes C, Albarracin D. Recalling and Intending to Enact Health Recommendations: Optimal Number of Prescribed Behaviors in Multibehavior Messages. Clin Psychol Sci. 2017;5(5):858-865.
- Tagliabue E, Fargnoli MC, Gandini S, et al. MC1R gene variants and non-melanoma skin cancer: a pooled-analysis from the M-SKIP project. Br J Cancer. 2015;113(2):354-363.
- Pasquali E, Garcia-Borron JC, Fargnoli MC, et al. MC1R variants increased the risk of sporadic cutaneous melanoma in darker-pigmented Caucasians: a pooled-analysis from the M-SKIP project. Int J Cancer. 2015;136(3):618-631.
- Smit AK, Collazo-Roman M, Vadaparampil ST, et al. MC1R variants and associations with pigmentation characteristics and genetic ancestry in a Hispanic, predominately Puerto Rican, population. Sci Rep. 2020;10(1):7303.
- Hardt O, Nader K, Nadel L. Decay happens: the role of active forgetting in memory. Trends Cogn Sci. 2013;17(3):111-120.
- Dermatology AAo, 2012. https://www.aad.org/public/. Accessed 09/28/2017.
- Lacson JCA, Kim Y, Roetzheim RG, Sutton SK, Vadaparampil ST, Kanetsky PA. Predictors of genetic risk recall among the participants of a randomized controlled precision prevention trial against melanoma. Genet Med. 2023;25(4):100005.
- Powe BD. Cancer fatalism among elderly Caucasians and African Americans. Oncol Nurs Forum. 1995;22(9):1355-1359.
- Steidel AGL, Contreras JM. A new familism scale for use with Latino populations. Hispanic J Behav Sci. 2003;25(3):312-330.
- Glanz K, Yaroch AL, Dancel M, et al. Measures of sun exposure and sun protection practices for behavioral and epidemiologic research. Arch Dermatol. 2008;144(2):217-222.
- Zhang Z. Variable selection with stepwise and best subset approaches. Ann Transl Med. 2016;4(7):136.

