Lipogenic Phenotypes
Project Summary:
Altered metabolism is an emerging hallmark of cancer. It is well accepted that malignant tumors consume significantly more glucose than surrounding normal tissue. This elevated glucose consumption coupled with poor vascular perfusion leads to a highly acidic microenvironment, with extracellular pH values a low as 6.5. Adaptations to this profoundly acidic microenvironment are necessary for tumor cells to survive and transition from an avascular pre-invasive tumor to a malignant invasive phenotype. Previously we have observed that one of the major survival mechanisms adopted by tumor cells is chronic activation of autophagy. This adaptation appeared to be necessary for the survival of tumor cells under low pH as inhibition of autophagy resulted in significant cell death under low pH, compared to normal pH, conditions. Further studies from our lab have demonstrated that cells that are adapted to grow under chronic exposure to acidity redistribute their lysosomes and decorate cell surface with lysosomal proteins, such as LAMP-2, and that this re-distribution protects the plasma membrane from acid-mediated cytotoxicity. We also noted that growth in acidic conditions leads to dramatic increases in cytoplasmic lipid droplets (adiposomes) which are dynamic organelles with cores of cholesterol esters (CE) and triacylglycerides (TAGs), surrounded by a shell comprised of polar lipids and proteins like the perilipins (Figure).
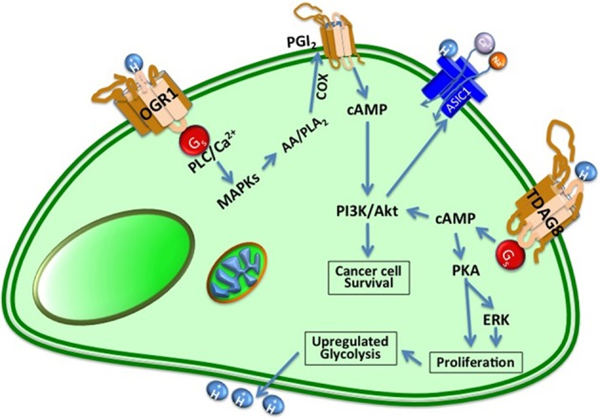
Figure 1: How acid signal is transduced in cells
It is believed that adiposomes function as crucial metabolic hubs by playing central roles in energy and membrane metabolism and production of signaling molecules. Although their biogenesis is well-studied in liver and adipocytes, it is not known why this phenotype is prevalent in cancer, or how and why it is induced by acidic conditions. Since accumulation of lipids as adiposomes occurs rapidly with acid exposure (as early as 6 hours) and can be reversed under neutral pH suggesting that this event dependent on the continuous presence of signal and it is likely that lipogenesis is dependent on continuous signaling from acid sensing plasma membrane receptors (OGR1, TDAG8, GPR1 & G2A) or acid sensing ion channels (ASICs) (Figure). We are currently investigating the role of acid “sensors” by generating CRSPR/Cas9 knock outs of major receptors and expect to unravel the downstream mediators of pH induced receptor activation and subsequent adiposome formation. The acid-induced lipogenic phenotype persists even with de-lipidated serum, indicating that the source of these lipids is de-novo and endogenous. In addition, we are currently employing stable isotope labeling (13C) to identify the mechanism and carbon source of the acid-induced lipogenic phenotype.
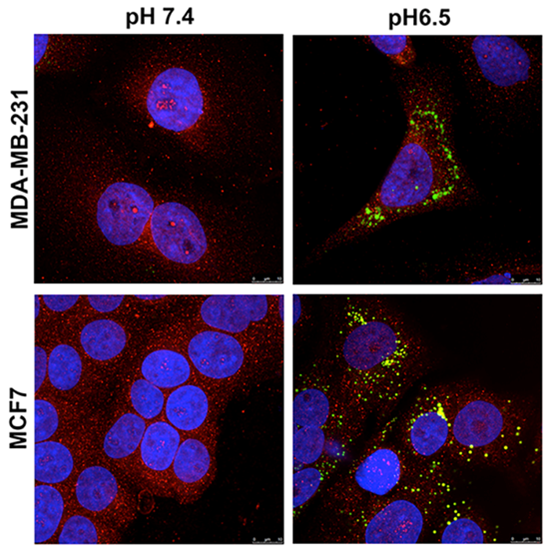
Figure 2: Acid-induced Lipogenic Phenotype
Project Members
Smitha Ravindranadhan Pillai, PhD
Research Scientist II/Project Lead
